Short Answer Questions
Question.1 Draw the electron dot structure of ethyne and also draw its structural formula.
Answer.

Question.2 Write the names of the following compounds.
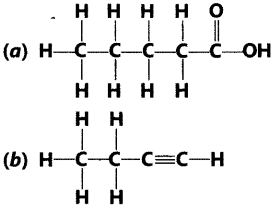
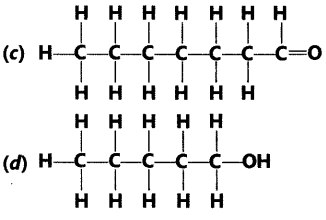
Answer.
(a) Pentanoic acid (b) 1-Butyne
(c) Heptanal (d) 1-Pentanol
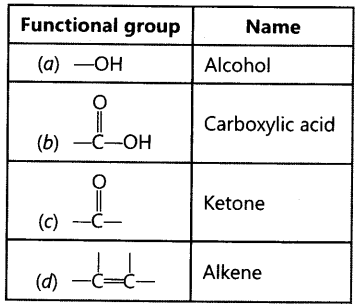
Question.3 Identify and name the functional groups present in the following compounds.
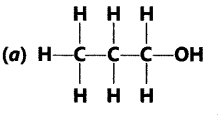
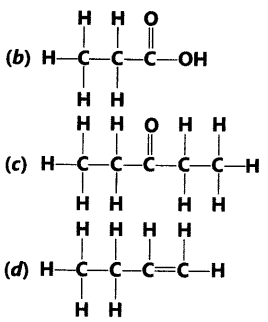
Answer.
Question.5 Why detergents are better cleansing agents than soaps? Explain.
Answer. It is because detergents form lot of lather even with hard water.
Hard water contains Ca2+ and Mg2+ ions which react with soap to form insoluble salts of calcium and magnesium called scum and soap goes waste. Detergents do not form insoluble compounds with Ca2+ and Mg2+ ions, therefore, these are more effective.
Question.6 Intake of small quantity of methanol can be lethal. Comment.
Answer. Methanol is oxidised to methanal in liver.’ Methanal is highly reactive and good reducing agent. It causes protoplasm to coagulate. It also affects optic nerve and leads to blindness.
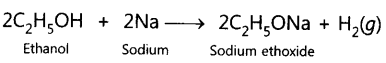
Question.7 A gas is evolved when ethanol reacts with sodium. Name the gas evolved and also write the balanced chemical equation of the reaction involved.
Answer. Hydrogen gas is evolved.
Question.8 Catenation is the ability of an atom to form bonds with other atoms of the same element. It is exhibited by both carbon and silicon. Compare the ability of catenation of the two elements. Give reasons.
Answer. Carbon shows catenation to large extent as compared to silicon as well as any other element due to smaller size of carbon. C—C bond is stronger than Si-Si bond because Si is larger in size, forms wea’ker bond.
Question.9 Match the reactions given in Column (A) with the names given in Column (B).
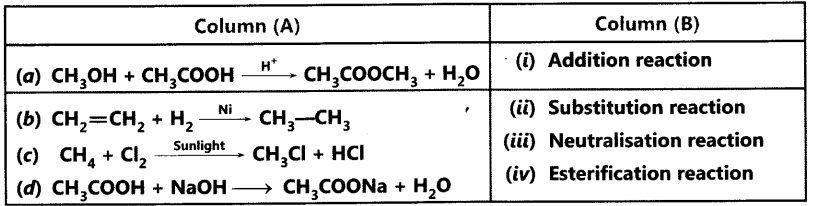
Answer.
(a) – (iv) is esterification reaction because ester is being formed from carboxylic acid and alcohol.
(b) – (i) is addition reaction as hydrogen is being added.
(c) – (ii) is substitution reaction because hydrogen of methane is being substituted by
chlorine atom.
(d) – (iii) is neutralisation reaction because acetic acid reacts with sodium hydroxide to form salt and water.
6. What is a homologous series? Explain with an example.
A homologous series is a series of compounds, which has the same functional group. This also contains similar general formula and chemical properties. Since there is a change in the physical properties, we can say that there would be an increase in the molecular size and mass.
For example, methane, ethane, propane, butane, etc. are all part of the alkane homologous series. The general formula of this series is CnH2n+2. Methane CH4 Ethane CH3CH3 Propane CH3CH2CH3 Butane CH3CH2CH2CH3. It can be noticed that there is a difference of −CH2 unit between each successive compound.
7. How can ethanol and ethanoic acid be differentiated on the basis of their physical and chemical properties?
Solution:
| Ethanol | Ethanoic acid |
| Does not react with sodium hydrogen carbonate | Bubbles and fizzes with hydrogen carbonate |
| A good smell | Smells like vinegar |
| No action in litmus paper | Blue litmus paper to red |
| Burning taste | Sour taste |
8. Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?
Solution: Micelle formation takes place because of the dirt particles in water and clean water. There are two mediums that are involved: one is pure water and the other being dirt (also called as impurities). The soap also has two mediums:
(i) organic tail and
(ii) ionic head
So the organic tail mixes and dissolves with the dirt whereas the oil or grease and ionic head dissolves and mixes with the water. Therefore, when the material to be cleaned is removed from the water, the dirt is taken off by the soap molecules in the water. Hence, the soap cleans by forming closed structures by the mutual repulsion of the micelles (positively charged heads).


