Laws of Chemical Combination:
The process of combination of two or more elements to form new compounds is governed by certain laws called laws of chemical combination. These are:
1. Law of conservation of mass.
2. Law of constant proportions.
Law of conservation of mass
- According to the law of conservation of mass, matter can neither be created nor destroyed in a chemical reaction. It remains conserved.
- Mass of reactants will be equal to the mass of products.
Law of constant proportions
- A pure chemical compound contains the same elements combined together in a fixed proportion by mass is given by the law of definite proportions.
- For e.g., If we take water from a river or from an ocean, both has oxygen and hydrogen in the same proportion.
Atoms
An atom is the defining structure of an element, which cannot be broken by any chemical means.
The atomic symbol has three parts:-
- The symbol X: the usual element symbol
- The atomic number A: equal to the number of protons
- The mass number Z: equal to the total number of protons and neutrons in an element.
Dalton’s Atomic Theory
- The matter is made up of indivisible particles known as atoms.
- The properties of all the atoms of a given element are the same, including mass. This can also be stated as all the atoms of an element have identical mass and chemical properties; atoms of different elements have different masses and chemical properties.
- Atoms of different elements combine in fixed ratios to form compounds.
- Atoms are neither created nor destroyed. The formation of new products (compounds) results from the rearrangement of existing atoms (reactants) in a chemical reaction.
- The relative number and kinds of atoms are constant in a given compound.
Atomic Mass
- Atomic mass is the total of the masses of the electrons, neutrons, and protons in an atom, or in a group of atoms, the average mass.
- Mass of an atomic particle is called the atomic mass.
- This is commonly expressed as per the international agreement in terms of a unified atomic mass unit (AMU).
- It can be best defined as 1/12 of the mass of a carbon-12 atom in its ground state.
![]()
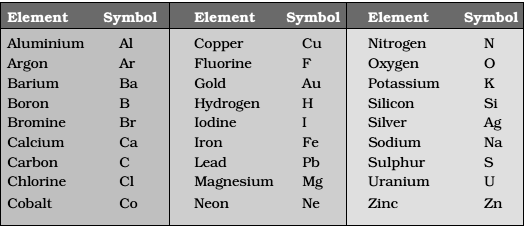
Names of Atoms or Elements and Their Symbols:
- IUPAC (International Union of Pure and Applied Chemistry) approves names of elements.
- The abbreviation used for lengthy names of elements are termed as their symbols.
- The symbol of an element is formed by writing only the first letter or first letter followed by the second or some other letter of English name or Latin name of the element.
- While writing a symbol, the first letter is always capital and the second is always small.
- Symbols used for some common elements are given below:
Molecules:
A group of two or more than two atoms of the same or different elements that are chemically bonded together is called a molecule.
For example: Two atoms of hydrogen (H2) and one atom of oxygen (O2) react with each other and form one molecule of water.
Atomicity:
The number of atoms present in a molecule of an element or a compound is known as its atomicity.
For example, atomicity of oxygen (O2) is 2 while atomicity of ozone (O3) is 3.
Ions:
It is an electrically charged atom or group of atoms. It is formed by the loss or gain of one or more electrons by an atom.
Ions are of two types:
(i) Cation: It is positively charged ion and is formed by the loss of one or more electrons from an atom
For example: sodium atom, loses one electron to form a sodium ion Na
Na – e− → Na+
(ii) Anion: It is a negatively charged ion and is formed by the gain of one or more electrons by an atom.
For example a chlorine atom gains one electron to form a chloride ion Cl−.
Cl + e− → Cl−
Valency:
It is defined by the combining power (or capacity) of an element.
Depending on their valency, elements can be classified as following:
(i) Monovalent cation: Having cationic valency of 1.
For example: Sodium ion (Na+ ). Potassium ion (K+ ), Hydrogen ion (H+ ).
Monovalent anion: Having anionic valency of -1.
For example: Chloride ion (Cl-), Bromide ion (Br-)
(ii) Divalent cation: Having cationic valency of 2.
For example: Magnesium ion (Mg2+), Ferrous ion (Fe2+).
Divalent anion: Having anionic valency of −2.
For example: Oxide ion (O2−), Sulphide ion (S2−).
(iii) Trivalent cations: Having cationic valency of 3.
For example: Aluminium ion (Al3+), Ferric ion (Fe3+).
Trivalent anion: Having anionic valency of -3.
For example: Nitride ion (N-3), Phosphate ion (PO43−) etc.
Ionic compounds: chemical formula
Steps :
(i) While writing the chemical formulae for compounds, write the constituent elements with their valencies written down the respective elements.
(ii) Then crossover the valencies of the combining atoms as shown in the following examples.
Molecular Mass:
The number of times a molecule of a compound is heavier than the 1/12 of the mass of C-12 atom, is known as its molecular mass.
The molecular mass is equal to the sum of the atomic masses of all atoms present in one molecule of the substance.
For example, Molecular mass of H2O= 2 × Mass of one H-atom + Mass of one O-atom = 2 ×1 + 16 = 18 u.
Formula unit mass:
It is the sum of the atomic masses of all atoms in a formula unit of a compound.
Formula unit mass is used for those substances whose constituent particles are ions.
For example, formula unit mass of ionic NaCl = 23 + 35.5 = 58.5 u.
Mole Concept:
- Mole: A collection of 6.023 × 1023particles is named as one mole.
1 mole = 6.023 × 1023 particles = Mass of 1 mole particles in grams
- The mass of 1 mole particles is equal to its mass in grams.
1 mole atoms = gram atomic mass
1 mole molecules = gram molecular mass
- Avogadro’s constant or Avogadro’s number:
The number of particles present in one mole (i.e. 6.023 x 1023 particles) is called Avogadro’s number or Avogadro’s constant.



