Introduction:
Dalton’s atomic theory in suggested that an atom was indivisible. However, the discovery of two fundamental particles named as electrons and protons, inside the atom, led to the failure of Dalton’s atomic theory.
Fundamental particles of an atom:
Three particles; electron, proton and neutron from which an atom is consisted of, are called fundamental particles of an atom or sub atomic particles.
Discovery of Electron:
By J. J. Thomson in 1897.
He carried a cathode ray experiment in which observed a stream of negatively charged particles coming out of cathode towards the anode. These particles were named as electrons.

Thomson’s model of an atom proposed that:
- An atom is a uniform sphere of positive charges (due to presence of protons) as well as negative charges (due to presence of electrons).
- Atom as a whole is electrically neutral because the negative and positive charges are equal in magnitude.
Limitations of Thomson’s Model:
- It failed to explain how protons and electrons were arranged in atom so close to each other.
2. Rutherford’s model of an atom
This was based on an experiment in which α – particles were bombarded on a thin gold foil.
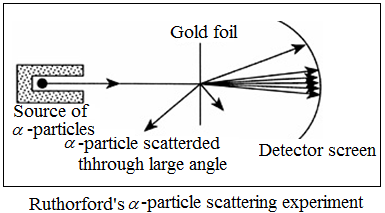
Observations of Rutherford’s α-particle scattering experiment are:
(a) Most of the α – particles passed without any hindrance.
(b) Some of the α – particles deflected from their original path at a noticeable angle.
(c) Very few of the α – particles bounced back at their original path.
Drawbacks of Rutherford Model
(a) According to Rutherford’s Model, electron revolves round the positively charged nucleus which is not expected to be stable. But a charged particle in an accelerated motion along a circular path would continuously undergo loss of energy and finally would fall into nucleus. This makes an atom unstable while atoms are quite stable.
(b) Rutherford model could not solve the problem of atomic mass of atom as it proposed only the existence of protons in the nucleus.
Bohr’s Model of Atom (By Neils Bohr in 1913)
This model of atom states that:
- An atom consists of heavy positively charged nucleus. The whole mass of the atom is concentrated in the nucleus.
- The electrons in an atom revolve around the nucleus in definite circular paths called orbits or energy level.
- Each energy level is associated with definite amount of energy.
- The change in energy takes place when electron jumps from one energy level to another energy level.
Arrangement of electrons in an atom:
The arrangement of electrons in various shells (energy levels) of an atom of the element is known as Electronic configuration.
The Maximum number of electrons that could be put in a particular shell, i.e., energy levels, was given by Bohr and Bury.
According to Bohr-Bury Scheme:
- The maximum number of electrons that can be accommodated in any energy level is given by 2n2where n = 1, 2, 3, 4, …. (for K, L, M, N…..)
- The maximum number of electrons in the outermost orbit will be 8 electrons even if it has capacity to accommodate more electrons.
- Electrons, are not accommodated in a given shell. Unless earlier shells are filled, that is stepwise filling of shells is followed.
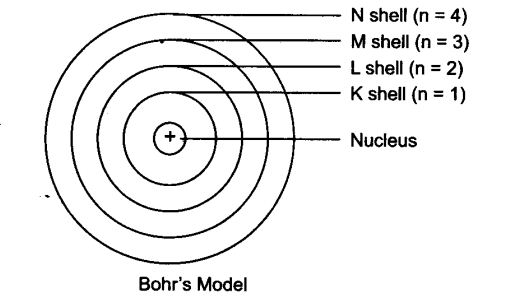
Orbit
The circular path around the nucleus is called orbit, energy level or shell. Energy level are represented by English alphabets: K, L, M, N, …. and so on.
Therefore,
- 1st orbit is denoted by K
- 2nd orbit is denoted by L
- 3rd orbit is denoted by M, and so on.
Distribution of Electrons in a Orbit or Shell
The distribution of electrons in an orbit can be obtained by using formulae 2n 2 where ‘n’ is number of that orbit.
For example:
Number of electrons in K-shell i.e. in 1st orbit
Here n = 1
Therefore, 2n2 = 2 × 12 = 2
Thus, maximum number of electrons in K-shell, i.e. 1st shell = 2
Number of electrons in L-shell, i.e. in 2ndorbit
Here n = 2, therefore,
2n2 = 2 × 22 = 8
Thus, maximum number of electrons in L-shell = 8
In similar way maximum number of electrons in any shell can be calculated.
Valence Electrons
The electrons present in the outer most shell of an atom are known as valence electrons. These electrons determine the valency of an atom.
Valency
Valency is the combining capacity of an atom, i.e. their tendency to react and form molecules with atoms of the same or different elements.
For the atoms having valence electrons less than or equal to 4, valency is same as that of the number of valence electrons in that atom.
For example, valency of Magnesium (2, 8 , 2) = 2
If number of valence electrons exceed 4, then valency = 8 – Number of valence electrons.
For example, valency of Oxygen (2, 6) = 8 – 6 = 2.
Valency of atoms with 8 valence electrons is zero as they have fully filled valence shell and cannot gain or lose electrons to form, molecules or compounds.
Atomic number (Z)
Atomic number of an element is equal to the number of protons present in the nucleus of an atom.
Since an atom is electrically neutral, thus number of protons and number of electrons are equal, therefore:
Atomic number (z) = number of protons = number of electrons.
Mass Number (A)
The total number of the protons and neutrons present in the nucleus of an atom is called mass number. It is denoted by A.
The protons and neutrons together are called nucleon.
The number of neutrons present in the nucleus of an atom is rperesented by N.
Isotopes
Atoms of the same element having same atomic number but different mass numbers are known as Isotopes.
For example:
Applications of Isotopes
- Isotope of uranium is used as a fuel in nuclear reactor
- Isotope of cobalt is used in treatment of cancer
- Isotope of iodine is used in treatment of goitre.
Isobars
Atoms of different elements having same mass numbers are known as Isobars,
For example:
Potassium, K and Argon. Ar both have the mass numbers equal to 40.
NCERT Solutions from Textbook
Question 1. Which of the following are matter?
Chair, air, love, smell, hate, almonds, thought, cold, cold-drink, smell of perfume.
Answer: Chair, air, almonds, and cold-drink.
Question 2. Give reasons for the following observation:
The smell of hot sizzling food reaches you several meters away, but to get the smell from cold food you have to go close.
Answer: The smell of hot sizzling food reaches severed meters away, as the particles of hot food have more kinetic energy and hence the rate of diffusion is more than the particles of cold food.
Question 3. A diver is able to cut through water in a swimming pool. Which property of matter does this observation show?
Answer: A diver is able to cut through water in a swimming pool. This shows that the particles of water have intermolecular space and has less force of attraction.
Question 4. The mass per unit volume of a substance is called density.
(density = mass/volume).
Arrange the following in order of increasing density: air, exhaust from chimneys, honey, water, chalk, cotton and iron.
Answer: Increasing density:
air < exhaust from chimneys < cotton < water < honey < chalk < iron.
Question 5. (a) Tabulate the differences in the characteristics of states of matter.
(b) Comment upon the following: rigidity, compressibility, fluidity, filling a gas container, shape, kinetic energy and density.
Answer: (a) Difference in the characteristics of 3 states of matter.

b) Comment on:
(i) Rigidity: The tendency of a substance to retain/maintain their shape when subjected to outside force.
(ii) Compressibility: The matter has intermolecular space. The external force applied on the matter can bring these particles closer. This property is called compressibility. Gases and liquids are compressible.
(iii) Fluidity: The tendency of particles to flow is called fluidity. Liquids and gases flow.
(iv) Filling of a gas container: Gases have particles which vibrate randomly in all the directions. The gas can fill the container.
(v) Shape: Solids have maximum intermolecular force and definite shape.
Whereas liquids and gases takes the shape of container.
(vi) Kinetic energy: The energy possessed by particles due to their motion is called kinetic energy. Molecules of gases vibrate randomly as they have maximum kinetic energy.
(vii) Density: It is defined as mass per unit volume, the solids have highest density.
Question 3. Give reasons :
(a) A gas fills completely the vessel in which it is kept.
Answer: The molecules of gas have high kinetic energy due to which they keep moving in all directions and hence fill the vessel completely in which they are kept.
(b) A gas exerts pressure on the walls of the container.
Answer: A gas exerts pressure on the walls of the container because the molecules of the gas are in constant random motion due to high kinetic energy. These molecules constantly vibrate, move and hit the walls of the container thereby exerting pressure on it.
(c) A wooden table should be called a solid.
Answer: The molecules/particles of wooden table are tightly packed with each
other, there is no intermolecular space, it cannot be compressed, it cannot flow, all these characteristics are of solid. So wooden table should be called a solid.
(d) We can easily move our hand in air but to do the same through a solid block of wood we need a karate expert.
Answer: We can easily move our hand in air but to do the same through a solid block of wood we need a karate expert. It is because the molecules of air has less force of attraction between them and a very small external force can separate them and pass through it. But in case of solids, the molecules have maximum force of attraction, the particles are tightly bound due to this force. Hence large amount of external force is required to pass through solid.
NCERT Solutions Textbook



