Elements are classified as metals and non-metals based on different properties. The properties of metals and non-metals are given in the form of table below-
| Metals | Non-metals |
| Metals are lustrous, that is, they have a property to shine. | They are not lustrous, that is, they do not have shining surface. except, graphite and iodine |
| All metals exist as solids except mercury which is liquid at room temperature. | They are generally soft, except diamond. |
| They can be drawn into wires, this is known as Ductility. | They are non-ductile. |
| Metals can be converted into sheets, this is known as Malleability, except mercury | They are non-malleable |
| They are good conductors of electricity and heat. Except Lead and mercury. | They are poor conductors of electricity and heat. Exception-graphite is good conductor of electricity |
| They have high density and high melting point. Exception-sodium and potassium have low melting points | They have low density compared to metals and low melting point except Diamond which has high melting point |
Chemical Properties of Metals
- Metals react with air or oxygen to form metal oxide.
For Example, Copper reacts with oxygen to form copper oxide.
Metal + O2 → Metal oxide
2Cu + O2 → 2CuO
4Al + 3O2 → 2Al2O3
- Oxides of metals can react with both acids and bases to produce salt and water. Such oxides are known as Amphoteric Oxides.
Al2O3 + 6HCl → 2AlCl3 + H2O

- Metals also reacts with water to form metal oxide. Metal oxide in turn can react with water to form metal hydroxide. For Example
2Na + 2H2O → 2NaOH + 1H2
2Al + 3H2O → Al2O3 + 3H2
- Metals also reacts with dilute acids to form salt and hydrogen. For example, magnesium reacts with dilute hydrochloric acid to form magnesium chloride and hydrogen.
Metal + Acid → Metal Salt + Hydrogen
Mg + 2HCl → MgCl2 + H2
Chemical Properties of Non-metals
- Non-metals reacts with oxygen to form non-metal oxide.
Non-metal + Oxygen → Non-metal oxide
C + O2 → CO2
- Non-metals do not react with water and acids to evolve hydrogen gas.
- Non-metals can react with salt solution; more reactive element will displace the less reactive non-metal.
2 NaBr (aq) + Cl2(aq) → 2NaCl (aq) + Br2 (aq)
- Non-metals can also react with hydrogen to form hydrides.
H2(g) + S(l) → H2S(g)
Reactivity Series
The series in which metals are arranged in the decreasing order of reactivity, it is known as Reactivity Series.
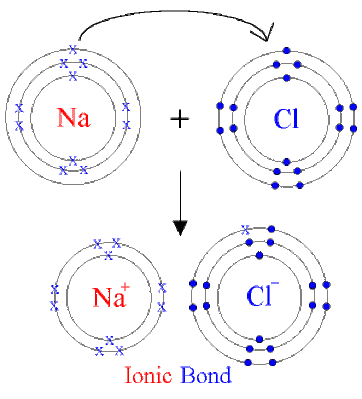
Ionic Compounds
Compounds formed due to the transfer of electrons from a metal to a non-metal are known as Ionic Compounds.
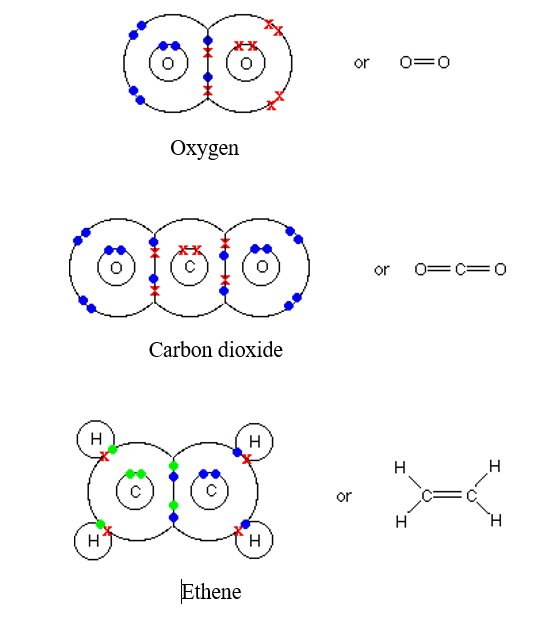
Covalent Bond
 [/vc_column_text]
[/vc_column_text]
Refining of Metals
Refining of impure metal is done using electrolytic refining. Impure copper is used as anode and strip of pure copper is used as Cathode. Acidified copper sulphate is used as electrolyte. When electric current is passed through this, impure metal from the anode gets deposited in the electrolyte solution, whereas pure metal from the electrolyte is deposited at cathode.
Deposition of insoluble residue formed from the dissolution of anode during commercial electrolysis.

Fig.2. Electrolytic refining
Corrosion
Metals when exposed to moist air for a long period of time, they become corroded. This is known as Corrosion. For Example, Silver reacts with moist air and becomes black in colour due to silver sulphide coating.
Iron + oxygen → Iron (III) oxide
Fe + O 2 → Fe2O3
Prevention of Corrosion
Rusting of iron can be prevented by oiling, galvanizing, painting, greasing etc.
To protect steel and iron from rusting, a thin layer of zinc are coated on them, this is known as Galvanization.
Alloy
Mixture of two or more metals or metal and non-metal is known as Alloy. For Example,
Brass is an alloy of copper and zinc.
Bronze in an alloy of copper and tin.
Solder is an alloy of lead and tin.
Amalgam is one metal is mercury.
ncert solutions

